Regulatory Intelligence: The Cornerstone of Successful Product Registration
In the fast-evolving world of pharmaceuticals, medical devices, and consumer health products, regulatory intelligence (RI) has become more than just a support function — it’s a strategic tool. Whether you are launching a new medicine, registering a medical device, or expanding into new markets, having the right regulatory insights can make the difference between a smooth approval process and costly delays.
What Is Regulatory Intelligence?
Regulatory Intelligence refers to the systematic collection, analysis, and interpretation of information about current and emerging regulations, policies, guidelines, and enforcement trends that may impact the development, registration, and marketing of products.
In simple terms, it’s about staying ahead of regulatory changes — knowing what authorities require today and what they are likely to require tomorrow.
Why Regulatory Intelligence Matters in Product Registration
- Ensures Compliance and Reduces Risk
Regulatory requirements vary across regions and are constantly changing. RI helps organizations stay compliant by providing up-to-date knowledge of regulatory expectations, minimizing the risk of non-compliance, delays, or product recalls. - Supports Faster Market Entry
With well-analyzed regulatory data, companies can design submission strategies that meet local authority expectations the first time — leading to faster approvals and earlier market access. - Enhances Decision-Making
Regulatory Intelligence provides actionable insights that help teams choose the best pathway for product registration — whether through full dossier submission, abridged applications, reliance mechanisms, or special access routes. - Optimizes Resource Planning
By anticipating regulatory challenges early, companies can allocate the right resources — time, budget, and personnel — to critical steps in the registration process. - Improves Competitiveness
Companies that invest in strong regulatory intelligence systems can respond faster to opportunities, such as tender requirements, regulatory harmonization initiatives, or new regional markets opening to certain product categories.
Key Components of Effective Regulatory Intelligence
To build a strong RI framework, organizations should focus on:
- Monitoring Sources
Constantly tracking updates from regulatory authorities (like TMDA, WHO, EMA, FDA, or TBS) and regional harmonization bodies (like EAC-MRH or SADC). - Data Analysis and Interpretation
Turning raw information into meaningful insights by comparing regulatory expectations, identifying trends, and forecasting potential changes. - Information Sharing
Disseminating insights across departments — from R&D to regulatory affairs and quality assurance — to ensure everyone is aligned with current requirements. - Documentation and Archiving
Maintaining a well-organized repository of regulations, guidelines, and correspondences that can be easily referenced during future submissions or audits.
Regulatory Intelligence in Practice
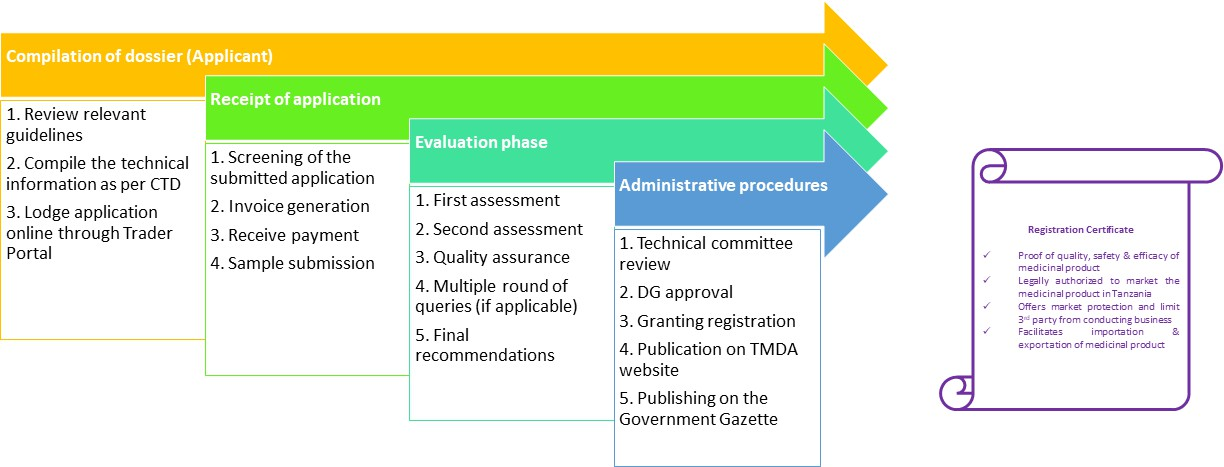
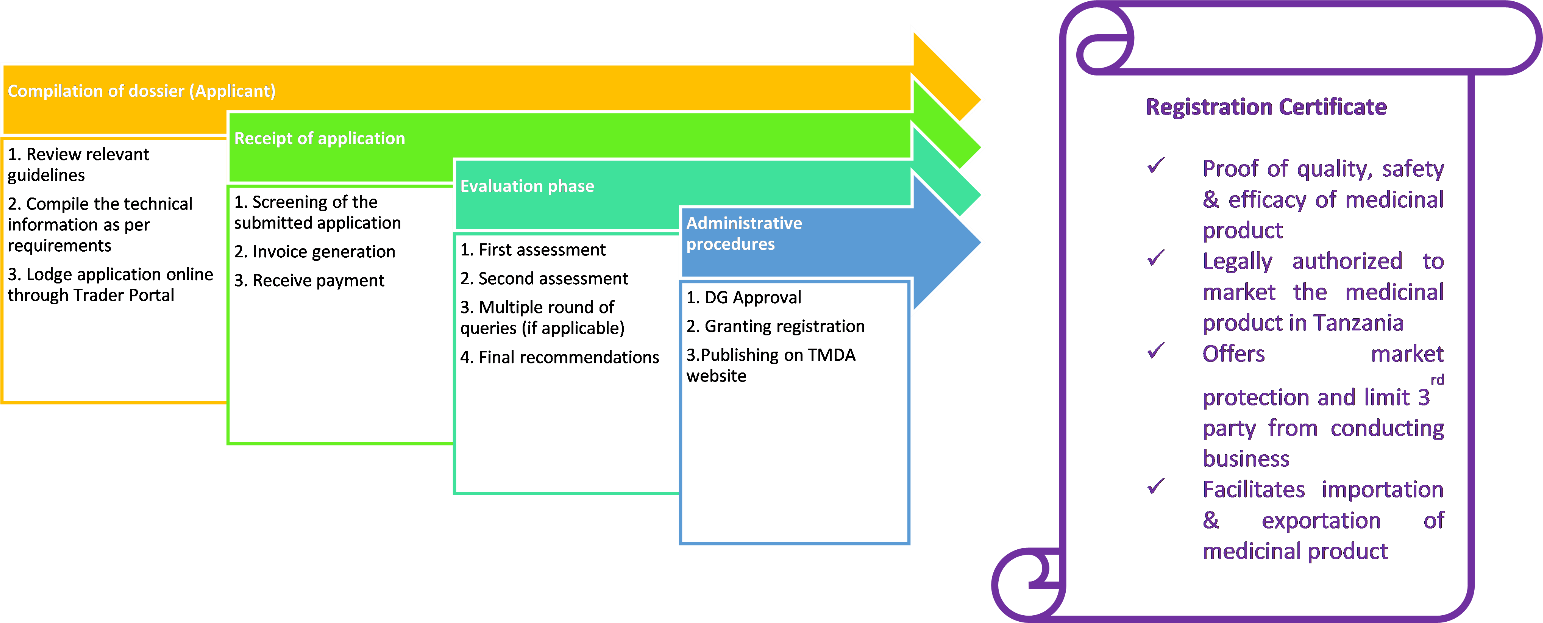
Consider a company planning to register a vaccine in Tanzania. By leveraging regulatory intelligence, the company can:
- Identify the most recent TMDA guidelines for vaccine registration.
- Understand stability data requirements specific to climatic zones (e.g., Zone IVb).
- Learn about waiver policies or reliance mechanisms for WHO-prequalified products.
- Prepare documents that meet local dossier structure and labeling expectations.
This approach not only saves time but also strengthens the credibility of the submission package.
How Alcare Consultancy Can Help
At Alcare Consultancy, we recognize that effective regulatory intelligence is the foundation of every successful product registration. Our team continuously monitors and interprets regulatory updates from regional and global authorities to support clients in:
- Product registration and renewal
- Variation submissions and post-approval changes
- Import and special permit applications
- Compliance with pharmacovigilance and quality requirements
We translate complex regulatory requirements into practical, actionable strategies that ensure your products reach the market efficiently and remain compliant throughout their lifecycle.
Conclusion
In today’s dynamic regulatory landscape, regulatory intelligence is not optional — it’s essential. Companies that proactively integrate RI into their registration planning gain a significant advantage in speed, compliance, and strategic foresight.
By partnering with experienced regulatory professionals, businesses can turn regulatory challenges into opportunities for growth and innovation.