The Role of the AMA in Streamlining African Product Registration
The African Medicines Agency (AMA) represents a transformative step in strengthening the regulation of medical products across the African continent. For decades, pharmaceutical manufacturers and distributors have faced fragmented regulatory requirements, lengthy approval timelines, and duplicated registration efforts across African markets. The AMA seeks to change this by promoting harmonized, transparent, and efficient regulatory systems that facilitate faster access to safe, effective, and high-quality health products.
As more African countries move toward adopting the AMA framework, understanding its roles, objectives, and practical benefits becomes essential for manufacturers, marketing authorization holders, and regulatory consultants. Equally important is the strategic support offered by expert consultancies—such as Alcare Consultancy—to help organizations navigate this evolving regulatory landscape.
The Mandate and Core Roles of the African Medicines Agency (AMA)
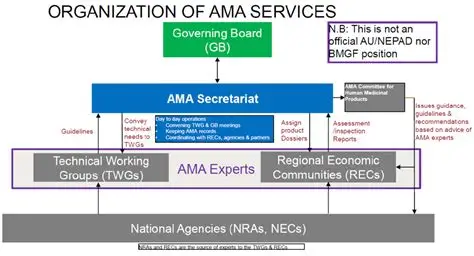
The AMA was established under the Treaty for the Establishment of the African Medicines Agency, endorsed by the African Union (AU). It builds upon earlier regional harmonization efforts such as the African Medicines Regulatory Harmonization (AMRH) initiative, which sought to align technical requirements among Regional Economic Communities (RECs) like EAC, SADC, and ECOWAS.
The AMA’s key roles include:
- Regulatory Harmonization Across Africa
AMA promotes the use of common technical documents, standardized guidelines, and shared regulatory assessment tools. This allows companies to submit a single, harmonized dossier that can be used across multiple member states, significantly reducing redundancy. - Capacity Building and Technical Support
AMA strengthens the capabilities of National Medicines Regulatory Authorities (NMRAs) through training, technical assistance, and collaborative assessments. This ensures consistency in decision-making and improves the overall regulatory quality in Africa. - Centralized Assessment and Mutual Recognition
Under the AMA framework, a product approved by one recognized authority (or through joint assessment) can be mutually recognized by participating countries, speeding up market entry. - Coordination of Pharmacovigilance and Safety Monitoring
AMA enhances regional pharmacovigilance systems by establishing data-sharing mechanisms and coordinating post-market safety activities among NMRAs. - Facilitating Access to Priority and Essential Medicines
The agency prioritizes products addressing public health needs—including vaccines, antimalarials, antiretrovirals, and innovative therapies—ensuring timely access for African populations.
Benefits of the AMA-Driven Registration Pathway
The AMA framework offers several advantages to both regulators and the pharmaceutical industry:
- Reduced Registration Timelines through joint reviews and shared assessments.
- Lower Regulatory Costs due to single dossier submission and minimized duplication.
- Improved Transparency with standardized evaluation criteria and digital submission platforms.
- Expanded Market Access by enabling companies to reach multiple countries with one regulatory effort.
- Stronger Post-Market Oversight through harmonized pharmacovigilance systems and collaborative inspections.
For businesses aiming to expand across Africa, AMA’s harmonized approach represents a strategic opportunity to accelerate product availability while ensuring regulatory compliance.
How Alcare Consultancy Helps Clients Optimize the AMA Registration Route
At Alcare Consultancy, we recognize that navigating the transition from fragmented national regulations to a harmonized AMA-driven system requires expert guidance and tailored strategy. Our services are designed to help clients leverage the efficiencies of the AMA framework while remaining compliant with both continental and national regulatory requirements.
- Regulatory Intelligence and Strategic Planning
Alcare Consultancy provides up-to-date insights on AMA developments, participating countries, and implementation timelines. We guide clients on how to align their registration strategies with current AMA frameworks and regional harmonization initiatives (e.g., EAC-MRH, SADC-MRH).
- Dossier Preparation and Harmonization Support
We assist in compiling Common Technical Dossiers (CTD) that meet AMA and local NMRA requirements. Our experts ensure that product data, labeling, and pharmacovigilance plans are optimized for submission under harmonized systems—reducing the need for reformatting or resubmission.
- Liaison with Regulatory Authorities
As part of our service, we act as a bridge between clients and regulatory authorities, ensuring clear communication, timely responses, and smooth follow-up throughout the joint review process.
- Pharmacovigilance and Post-Market Compliance
Alcare Consultancy provides Qualified Person for Pharmacovigilance (QPPV) services and establishes compliant safety monitoring systems in alignment with AMA’s post-authorization requirements. This ensures that safety data is properly managed across all participating markets.
- Training and Capacity Building
We offer targeted training for regulatory and quality teams to familiarize them with AMA procedures, documentation standards, and compliance expectations—empowering our clients to remain self-sufficient and compliant in the long term.
Looking Ahead: The Future of AMA and Its Impact on African Healthcare
As more African Union member states ratify the AMA treaty and operationalize their participation, the regulatory landscape in Africa is set to become more predictable, efficient, and innovation-friendly. This shift will not only accelerate the introduction of essential medicines but also encourage global manufacturers to invest in Africa’s healthcare ecosystem.
For organizations looking to register products across multiple African countries, early engagement with the AMA pathway—supported by expert regulatory partners like Alcare Consultancy—can result in faster approvals, wider market reach, and sustainable compliance.
Conclusion
The African Medicines Agency is ushering in a new era of regulatory collaboration and efficiency in Africa’s pharmaceutical sector. While the system is still evolving, forward-looking companies can already benefit from its harmonization goals by strategically planning their submissions and leveraging expert support.
Alcare Consultancy stands ready to assist manufacturers, importers, and distributors in optimizing their AMA registration journey—ensuring regulatory compliance, accelerating approvals, and contributing to better healthcare access across Africa.